
Among the different new materials with high temperature superconductivity, fullerene compounds play a significant part. Fullerenes are hollow-shaped carbon molecular aggregates the simplest and most symmetric of which is the C60 molecule composed of 20 hexagons and 12 pentagons, as on a football. Discovered in 1985 by Curl, Kroto and Smalley (Nobel Prize in 1996) [http://nobelprize.org/nobel_prizes/chemistry/laureates/1996/], this molecule with a diameter of 0.7 nanometres was quickly mass-produced, vaporizing graphite thanks to an electric arc. Isolated by chromatography, these molecules can form a solid with a cubic structure, where fullerenes occupy each corner and each face centre of the cube.
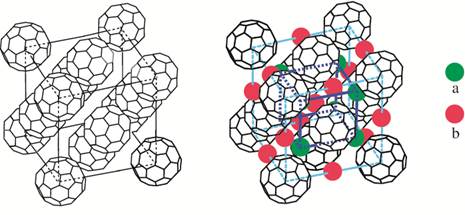
This solid composed of neutral molecules is insulating, but it is easy to insert atoms in the big intermolecular holes. It is hence possible to make AnC60 solids where n alkalines A ionize and give away their electrons to the C60 balls. The one composed of 3 alkalines for one fullerene is superconducting. For instance, Rb3C60 has a critical temperature Tc of 27 K (-246°C). If a strong pressure is applied to the compound Cs3C60, the Tc can even increase to 40 K.
The molecular structure of this compound gives it significant superconducting properties. Today, it is accepted that the interaction responsible for the electron pairing is located on the ball and is caused by the vibration modes of the ball. In other words, the electron pairs form on the ball using vibrations as in the BCS model and jump from ball to ball. In addition to that, since these materials are highly compressible, a pressure applied to reduce the distance between fullerenes enables to increase the metal characteristics and change the Tc. The same effect is observed at ambient pressure if the alkaline is replaced by another alkaline with a smaller ionic radius (Rb by K, for instance).
In order to try and increase the Tc, attempts to insert Cs (the biggest alkaline ion) were made, but it was only in 2008 that the synthesis of Cs3C60 compounds was achieved. The most surprising part is that the space between C60 is so big that the electrons are pinned to the balls and prevent them from relocating. At atmospheric pressure, the Cs3C60 compound is hence both insulating and magnetic. However, under a stronger pressure, the compound becomes a superconducting metal with a maximum Tc of 35 K (-238°C). This transition from an insulator to a metal (Mott transition) reminds of what happens in cuprates, pnictides, and heavy fermion compounds. This is why research activities on fullerene alkaline compounds have started their revival very recently.
